Waterborne Polymer Emulsions: Definition, Nature, Formation, and Classification
1. What is a Waterborne Polymer Emulsion?
A waterborne polymer emulsion is a dispersed system in which polymer particles, insoluble in water, are formed and stably distributed within an aqueous medium. In this system, the polymer does not exist in a dissolved state but as discrete particles of very small size, forming a colloidal system.
The polymer component determines fundamental properties such as mechanical strength, flexibility, deformability, and film-forming ability. Meanwhile, water acts as the dispersion medium, allowing the system to remain in a liquid state for convenient handling and application.
An important point is that a polymer emulsion is not simply “polymer in liquid form,” but a system composed of numerous individual polymer particles. Therefore, the material properties depend not only on the polymer itself but also on particle structure and the state of the entire dispersed system.

2. Polymer Emulsion as a Colloidal System
In essence, a polymer emulsion is a colloidal system in which very small polymer particles are dispersed in water at high concentration.
This state is maintained by a balance of interfacial forces. Polymer particles typically carry like charges or are surrounded by stabilizing layers, creating repulsive forces that prevent direct contact. When repulsion dominates, the system remains stable and dispersed.
However, changes in environmental conditions—such as pH, salt concentration, temperature, or mechanical forces—can disrupt this balance. As a result, particles approach each other and may aggregate, altering the material behavior.
This demonstrates that emulsion stability depends not only on the polymer itself but also on surface conditions and the surrounding environment.
3. Polymer Emulsion is Not a Solution
A common misconception is to treat polymer emulsions as polymer solutions.
In a solution, molecules are fully dissolved and exist independently within the medium. In contrast, in an emulsion, the polymer exists as discrete particles, and interactions between these particles determine the system’s properties.
Therefore, characteristics such as viscosity, flow behavior, processability, and stability are not intrinsic properties of the polymer alone but result from interactions among particles within the entire system.
4. Particle Structure and Its Significance
Each polymer particle in an emulsion may have a complex structure rather than being a simple homogeneous entity. Particle size, size distribution, and surface characteristics play critical roles in determining system behavior.
When particles are small and uniformly distributed, the system tends to be more stable and exhibits more consistent performance. Conversely, a broad size distribution or non-uniform surface structure may lead to issues such as viscosity variation or aggregation tendencies.
Thus, two emulsions containing the same type of polymer can exhibit entirely different performance in practice if their particle structures differ.
5. Formation of Polymer Emulsions
Waterborne polymer emulsions are not created by dissolving polymers but are formed directly in water through polymerization.
5.1. Nature of the Process
The polymer is generated in situ within the aqueous medium from initial monomers. During this process, polymer particles are formed and grow simultaneously with the chemical reaction.
This means that particle structure is determined during polymerization and directly influences the material properties.
5.2. Stages of Particle Formation
The process can be divided into three main stages:
- Initiation stage: formation of primary polymer particles
- Growth stage: particles grow as monomers diffuse into them
- Completion stage: the system reaches a stable state as monomers are nearly consumed
The number of particles, their size, and structure are determined at the early stage and maintained throughout the process.
5.3. Role of the Colloidal System in the Process
Throughout polymerization, the system behaves as a dynamic colloidal system. Factors such as temperature, agitation, monomer concentration, and stabilizers directly influence particle structure.
If not properly controlled, phenomena such as premature coagulation or non-uniform particle distribution may occur, leading to changes in material behavior.
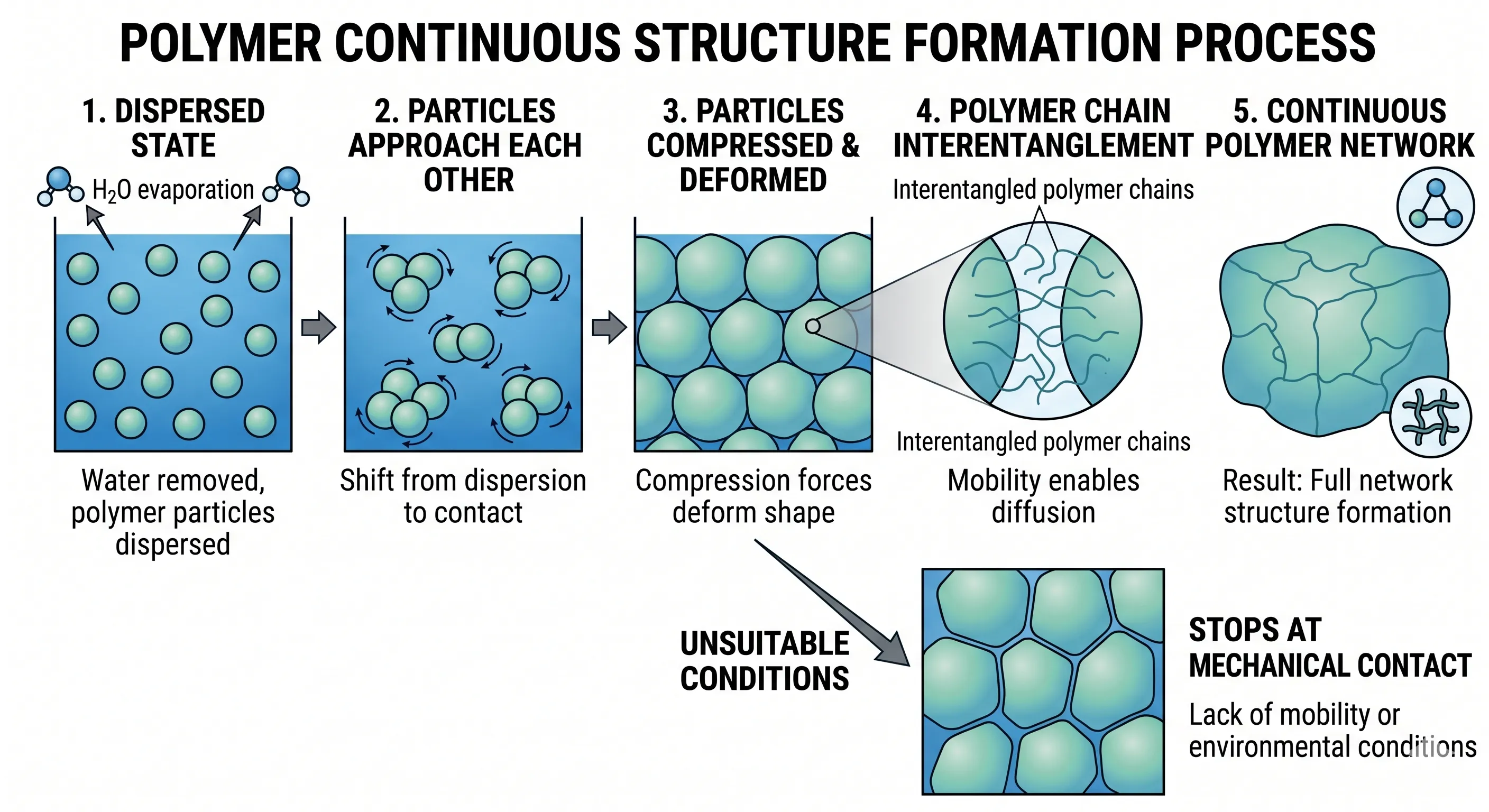
6. Formation of a Continuous Structure
When water is removed from the system, polymer particles move closer together and transition from a dispersed state to contact.
This process occurs in sequence:
- particles approach each other
- particles are compressed and deformed
- polymer chains interpenetrate
As a result, a continuous polymer network is formed.
This process only occurs completely when the polymer has sufficient mobility and environmental conditions are appropriate. Otherwise, the system may remain at a stage of mechanical contact without forming a fully developed structure.
7. Classification of Polymer Emulsions by Monomer Type
Polymer emulsions are commonly classified based on the primary monomers used:
- Vinyl and vinyl ester groups (e.g., vinyl acetate): soft and easily deformable
- Acrylic and methacrylic groups: wide tunability of properties
- Styrenic groups: more rigid, enhancing mechanical strength
- Diene groups: highly flexible but environmentally sensitive
- Nitrile groups: improved strength and internal interactions
- Halogenated groups: significantly alter surface properties
- Dispersed polyolefins: difficult to disperse but possess unique properties
- Copolymers: combine multiple monomers to optimize performance
- Functional monomers: strongly influence properties even at low concentrations
However, it should be noted that classification by monomer reflects only part of the system’s nature. Actual behavior also depends on particle structure and the polymerization process.
8. Relationship Between Monomer Structure and Material Behavior
Monomer structure directly affects:
- polymer chain mobility
- particle deformability
- ability to form continuous structures
- environmental resistance
However, the actual behavior of a polymer emulsion depends not only on monomers but also on how the system is formed and the resulting particle structure.
9. AMIBOND’s Approach to Polymer Emulsions
From a materials perspective, waterborne polymer emulsions are defined not only by monomer type but by the entire polymerization process.
At AMIBOND, polymerization is considered central to material design. The focus is on controlling the formation of polymer particles—from initiation and growth to the stable state of the system.
Currently, AMIBOND develops and operates polymerization systems based on vinyl acetate, copolymers, and acrylic systems, aiming to balance deformability, mechanical strength, and dispersion stability.
In practical production, the ability to consistently polymerize a specific system is more important than expanding into too many different systems, as particle structure formed during this process directly determines material behavior.

10. Conclusion
Waterborne polymer emulsions are complex material systems in which polymers exist as dispersed particles in water.
Material properties depend not only on the polymer itself but also on particle structure, stabilization mechanisms, and the polymerization process. Understanding this fundamental nature is essential for accurately evaluating and effectively applying these materials in practice.

